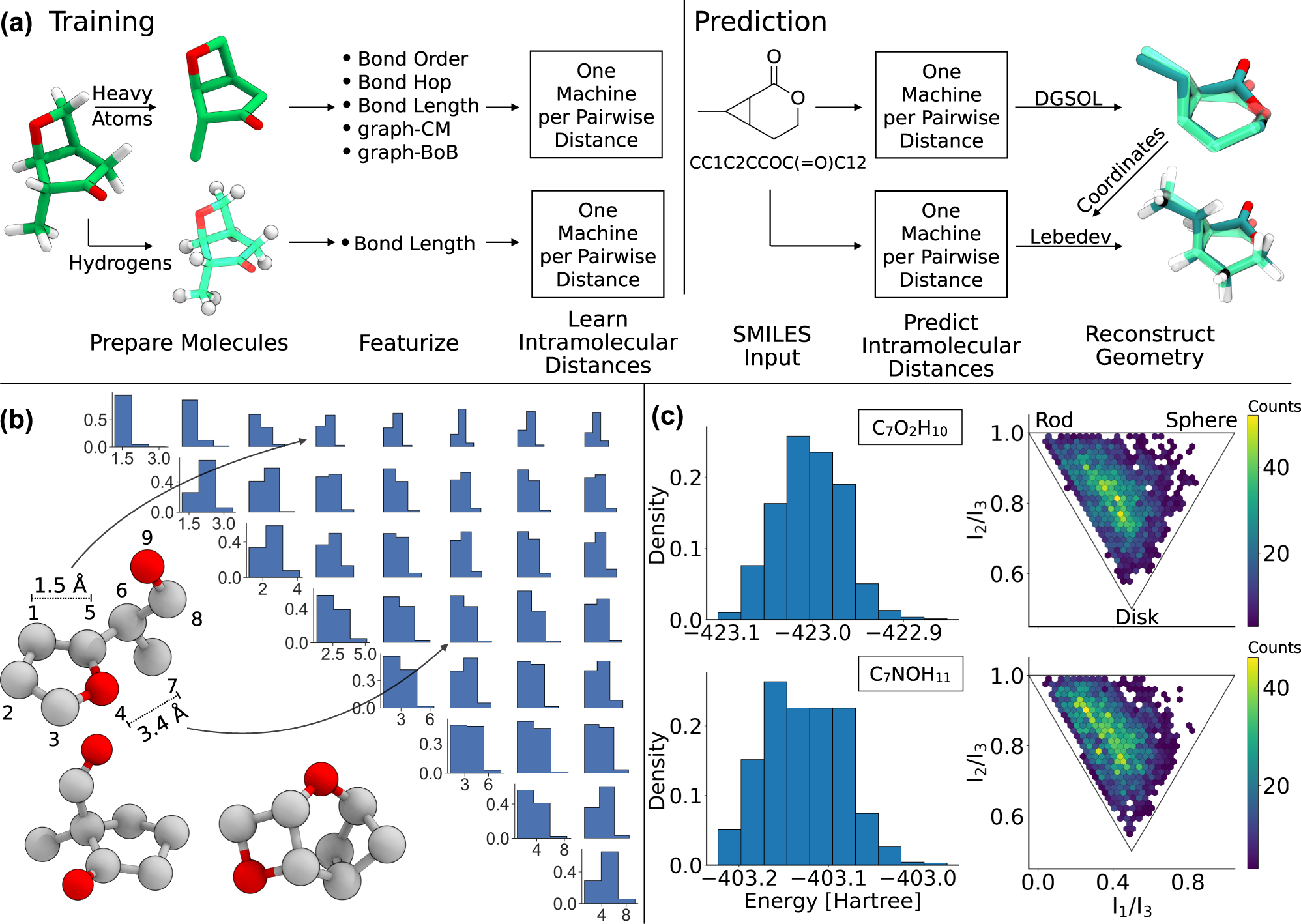
Machine learning based energy-free structure predictions of molecules, transition states, and solids | Nature Communications

All questions go with Model 4 , please label thank you in the Ltwis suettre for PCls and XeFe Model 4: Which Lewis Structure is Better? The two structures below could represent

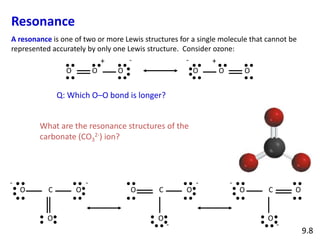
Explain why CO_(3^(2-) ion cannot be represented by a single Lewis structure. How can it be best represented?

Explain why CO_(3)^(2-) ion cannot be represented by a single Lewis structure. How can it be best represented?

Lewis Dot Structures Lewis Dot structures are used to represent the valence electrons of atoms in covalent molecules Dots are used to represent only the. - ppt download
Simplified T 1 triplet PES at the G3SX//M06-2X/aug-cc-pVTZ level of... | Download Scientific Diagram

Explain why CO_{3}^{2-} ion cannot be represented by a single Lewis structure. How can it be best represented?

Basic Concepts of Chemical Bonding Chapter 8. Three Types of Chemical Bonds Ionic bond Ionic bond –Transfer of electrons –Between metal and nonmetal ions. - ppt download